You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
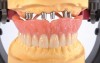
The removable denture market has undergone significant changes in recent years. New indications demand new solutions, for example, implant-supported hybrid dentures (Figure 1), which are said to achieve greater masticatory function and psychological satisfaction than conventional overdentures need to be able to withstand very different masticatory forces than did traditional full dentures.1 Laboratories that fabricate such restorations need teeth, denture acrylics, and other materials that meet increased demands regarding stability, function, and esthetics.
Since its introduction more than 70 years ago, polymethyl methacrylate (PMMA), or acrylic glass, has become the base material of choice for manufacturing denture bases and teeth.2 However, although the basic principle of acrylics processing —using a mixture of an MMA monomer and a PMMA powder to produce a paste, which then forms the basis of the denture—has remained unchanged, PMMA performance has been enhanced by the addition of fillers and processes designed to overcome shortcomings, including deficits in strength, abrasion, and stability. Further, new equipment and tools have brought new efficiency to standard processes, saving time that dental technicians can use more effectively to customize dentures, fine-tuning even the characteristic colors of the gingiva.
Requirements for Modern Dental Materials
Patients want to replace lost teeth in order to regain quality of life.3 While the most important purpose of dentures is the restoration of the chewing function for proper nutrition,4 patients also want dentures that are esthetically acceptable—ie, dentures that are not recognizable as such when they speak, smile, or chew. It is essential that dentures, too, provide oral comfort, if they are to be well accepted; therefore, the denture base should be matched or adapted perfectly to the jaw and retain its stable shape. Poorly fitted dentures and irregularities on the surface can cause bruising or irritation of the mucous membranes,5 or result in the denture’s not attaching properly, which can cause difficulty when speaking.4
In addition to the perfect occlusion on which function depends, both patients and their dentists are concerned that the prosthesis is designed to facilitate hygiene, as dentures need to be cared for properly to last and be enjoyable to wear. The accumulation of plaque and bacteria—particularly Candida albicans—is linked to surface roughness, so polishability is another important attribute in denture material.6 Also to support oral health, dentures must be made of biocompatible materials that will be well tolerated by the body, while being strong enough to stand up to the masticatory forces that enable wearers to enjoy and benefit nutritionally from a varied diet.
To keep client dentists and their patients happy, laboratories have requirements of their own. They need to be able to quickly and economically produce a durable, reliable product acceptable to all. This requires efficient processing of high-quality materials using equipment and methods that can support their ability to deliver dentures with the desired function and esthetics at an economical cost. This includes teeth that are easy to set up, and equipment that allows acrylics to be processed quickly.
Another challenge for laboratories is having the ability to accommodate the hybrid dentures that are gaining popularity. This means using denture materials in keeping with the unique masticatory demands of differing types of restorations and to allow secure bonding with other materials—eg, precious and non-precious metals—and otherwise be designed for modifications to be made if a patient’s oral cavity changes, eg, through the loss of additional teeth or alveolar ridge atrophy. Therefore material must be suited to re-linings and extensions and bond well to older material to form a single unit. In addition to being robust enough to function optimally within the mouth, modern dentures need to be able to withstand demands placed upon them outside the mouth—for example, if they are accidentally dropped during cleaning.
PMMA as a Denture Material
Currently, the most popular denture material is high-impact, heat-cured polymethyl methacrylate (PMMA).7 This glass-like thermoplastic resin is produced by polymerizing the methacrylic acid ester methylmethacrylate (MMA). During the process of radical polymerization, monomers are interlinked to produce a stable shape. It involves three steps summarized as: initiation, propagation, and termination.8,9,10 During initiation, the highly reactive radical starters bond with the first MMA molecule to trigger the chain reaction. During propagation, the first chain link reacts with additional MMA molecules and generates a new reactive radical and the chain continues to grow. Then, when the last link in the chain has reacted with another radical, the growth of the relevant chain terminates, transforming the formerly pliable substance into a hard, stable shape.
A recent study evaluating mechanical properties and dimensional stability of seven types of denture re-enforcement materials concluded that, among them, heat-cured polymethyl methacrylate with polyethylene fiber best accomplished the most critical requirements.11
While the vast majority of dentures manufactured worldwide are made of PMMA, other thermoplastic materials such as polyamide may be indicated in special circumstances—eg, in patients who are adversely affected by PMMA.12
PMMA-based Material Components8,9,10
Modern PMMA base materials for both artificial teeth and denture bases consist of a separate powder and a liquid formula—both of which have additional ingredients designed to compensate for problems associated with pure PMMA. Regardless of whether materials will be used in tooth production or in laboratories, the reliable processing of PMMA acrylics always depends on the careful adjustment of the powder/liquid formula. The core ingredients are liquid MMA, PMMA bead polymers, color pigments, initiators and cross-linking agents such as ethylene glycol dimethacrylate (EGDMA).
Liquid
The PMMA liquid consists of mainly of the monomer, methylmethacrylate (MMA), which, because it is thin, is able to partially dissolve its polymer and to combine with the powder into a homogeneous paste that can then be processed. During polymerization, however, MMA molecules combine to form PMMA chains, which act as the acrylic matrix, which allows the acrylic to fully cure. In addition to MMA, the liquid contains a stabilizer, a cross linker, and co-radical initiators. Stabilizers ensure that unpolymerized MMA, which is volatile, remains stable during storage. They capture unwanted, excess radicals by reacting with them before they can trigger polymerization. This increases the shelf life of the liquid. Very low concentrations of a peroxide such as hydroquinone are commonly used for this purpose. The cross-linking agents, EGDMA, ensure that the acrylic matrix is tightly linked. While MMA has one reactive group, cross-linking agents have at least two reactive groups. During the polymerization process, they can therefore bond with two chains and fix these in relation to each other. This results in a tight lattice structure that is resilient against chemicals, solvents, heat, and mechanical impact. Cross-linking agents offer the benefit of minimizing the risk of stress cracks in transparent acrylics, decreasing water sorption and solubility.9
Powder
The powder mainly consists of pre-polymerized PMMA or copolymer, which support the acrylic matrix as fillers to make the plastic dough. Because part of the acrylic has already cured, polymerization times, shrinkage, and residual monomer content are all reduced during laboratory processing.
The powder also contains color both organic and inorganic pigments and opacifiers, as well as the radical initiators needed to launch the polymerization process. Once the initiator breaks up to produce radicals, it starts the chain reaction, which may be precipitated by energy supplied by means of heat or light, or is generated by a chemical reaction due to the presence of a “co-radical initiator” in the liquid. Where, for example, the powder and liquid initiators combine in a barbituric acid initiation, they react with the oxygen in the ambient air and automatically trigger the polymerization process.9
Polymerization
The powder and liquid are mixed together in a 3:1 ratio, after which heat and pressure are applied. This process causes the double bonds to break and the occurrence of polymerization. Due to the free-radical addition reaction, there is no byproduct.10
The Use of Innovative Fillers in Denture Teeth13,14,15
To overcome the problem of abrasion with PMMA teeth, which persists despite improvements resulting from cross-linking, manufacturers have developed ceramic fillers (glass or silicate glass powder), which combine a highly cross-linked organic acrylic matrix with much smaller inorganic particles. These composites are designed to balance fracture resistance, color stability, abrasion resistance, and resistance against plaque formation, a delicate process, given that improvements in one aspect may diminish another aspect. For example, composite teeth in general offer good abrasion resistance, chemical stability and biocompatibility. However, there is concern with some that their inorganic fillers can be released from the matrix—eg, during polishing or chewing—and lead to a rougher surface, which in turn is associated with lower plaque resistance and faster discoloration of the teeth.
Processing Composite Teeth15
The process of manufacturing composite teeth involves many of the same steps used by laboratory technicians in their laboratories.
First, powder and liquid are mixed to form an acrylic paste, as described above, which is moulded. This prepolymerized dental material is then injected into temperature-resistant, computer-milled steel moulds in multiple, highly cross-linked layers.
High-quality composite tooth lines have a multi-layered structure with separate moulds used for each layer, which is individually heat-polymerized, starting with the incisal layer and followed by the dentin core and cervix of the tooth. This layered approach allows for adjustments in keeping with the requirements of each layer—eg, the incisal/enamel layer, which is exposed to high chewing loads, must be strongly resistant to abrasion, while the cervix, which forms the base of the tooth, should be less densely cross-linked and easier to partially dissolve in order to ensure an optimum bond with denture acrylics.
Processing Denture Bases15,16
Denture bases fabricated individually by laboratory technicians may utilize any of a number of different techniques and equipment. To precisely fit the denture to the individual patient’s oral cavity, a mould is first taken of the patient’s jaw. It is on this mould that the final prosthetic teeth are set in wax. The structure is then invested, after which the wax is boiled out. The resulting cavity is filled evenly with the denture acrylic.
Depending on the acrylic used, the patient indication, and the procedures applied by the laboratory, dental technicians commonly choose one of the four moulding techniques described below. Each has advantages and disadvantages
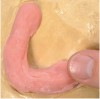
The pressing/packing technique (Figure 2) is typically used with heat-curing polymers to fabricate full dentures. The material is placed by hand into the flask, then hydraulically compacted using a press. This method is highly accurate and the equipment is inexpensive, but it is time- and material-intensive.
The pouring technique (Figure 3) is reserved for the fabrication of both full and partial dentures using cold-curing polymers. The denture material is cast into the mould without additional compaction. Although handling is simple and investment costs are low, this method leads to an initially higher residual monomer content, lower precision, and poorer material compaction compared to the injection method.
The injection technique is used both for heat-curing and cold-curing polymers used for full dentures. The denture material is placed into the flask at high pressure with an injection device. This method involves a high investment cost, but it offers high precision, low residual monomer content, and improved material compaction compared to the casting technique.
The injection moulding technique is specifically designed for use with thermoplastic resins and can be used with MMA-free plastics. This method is complex and requires special tools. The material is melted at a temperature exceeding 200 ̊C and injected into the flask at high pressure. Polymerization shrinkage can be minimized by injecting additional liquid material during the polymerization process.
Modern flasks/curvettes used with the injection methods are made from milled aluminum. They have several parts, which need to fit together precisely, interlock firmly, and be stable and temperature-resistant. They must be free from dents or deformations to produce precisely fitting dentures, and they must be specifically adapted to the injection device used.
Polymerization Techniques and Equipment15,16
Denture acrylic can be cured by heat (heat-curing polymers) or by triggering the radical initiator system (cold-curing polymers).
There are two different types of cold polymerization (autopolymerization) techniques used with cold-curing polymers. When an amine-peroxide initiator system is used, the initial reaction is triggered by mixing the powder and liquid at room temperature. The barbituric acid initiator system uses a pressure pot heated to 55 °C / 131 °F.
It is possible to inexpensively utilize the heat polymerization method using a standard cooking pot heated on a hot-plate. However, there are polymerization units available with hot water baths with time and temperature controls to allow programs to be run automatically, offering low susceptibility to error due to automatic time and temperature controls, with process length and temperature options.
Summary
Modern formulations of cured PMMA offer most of the ideal properties of a denture base, especially in that they are tasteless, odorless, chemically stable, biocompatible, natural looking, adhere to plastic, metal, and porcelain, easy to repair, and affordable. Further, they are transparent, scratch-resistant, durable, polishable and can be modified to achieve the esthetics patients demand, and they can be adapted to accommodate restorations in place and those that may be added later.
Modern equipment has facilitated more reliable and efficient moulding and curing processes for acrylics, both in dental laboratories and in industrial facilities, and CAD/CAM technology allows highly precise moulds to be manufactured for industrial tooth production.
References
1. Jain AR, Nallaswamy D, Ariga P, Philip JM. Full mouth rehabilitation of a patient with mandibular implant screw retained Fp-3 prosthesis opposing maxillary acrylic removable over-denture. Contemp Clin Dent. 2013;4(2):231-235.
2. Bonsor SJ, Pearson G. A Clinical Guide to Applied Dental Materials. Amsterdam ; Boston : Elsevier/Churchill Livingstone, 2013.
3. Pistorius J, Horn JG, Pistorius A, Kraft J. Oral health-related quality of life in patients with removable dentures. Schweiz Monatsschr Zahnmed. 2013;123(11):964-71; 955. [Article in English, German]
4. Knipfer C, Riemann M, Bocklet T, et al. Speech intelligibility enhancement after maxillary denture treatment and its impact on quality of life. Int J Prosthodont. 2014;27(1):61-69.
5. Budtz-Jorgensen E. Oral mucosal lesions associated with the wearing of removable dentures. J Oral Pathol. 1981;10(2):65–80.
6. Yamauchi M1, Yamamoto K, Wakabayashi M, Kawano J. In vitro adherence of microorganisms to denture base resin with different surface texture. Dent Mater J. 1990;9(1):19-24.
7. Balos S, Pilic B, Markovic D, et al. Poly(methyl-methacrylate) nanocomposites with low silica addition. J Prosthet Dent. 2014;111(4):327-334.
8. Tandon R, Gupta S, Agarwal SK. Denture base materials: From past to future. Indian J Dent Sci. 2010;2(2):33-39.
9. Bradna P. Polymerization: Synthetic polymers used in dentistry. http://che1.lf1.cuni.cz/html/Polymers.pdf. Accessed April 28, 2014.
10. Foulk C. Denture Polymers. http://quizlet.com/8258919/info. Accessed April 25, 2014.
11. Venkat R, Gopichander N, Vasantakumar M. Comprehensive analysis of repair/reinforcement materials for polymethyl methacrylate denture bases: mechanical and dimensional stability characteristics. J Indian Prosthodont Soc. 2013;13(4):439-449.
12. Leggat PA1, Kedjarune U. Toxicity of methyl methacrylate in dentistry. Int Dent J. 2003 Jun;53(3):126-31.
13. Shock absorbability and hardness of commercially available denture teeth. Kawano F, Ohguri T, Ichikawa T, et al. Intl J Prosthodontics. 2002;15(3).
14. Gürbüz Ö, Ünalan F, Kursoglu P. In vitro wear of denture teeth acrylic resin milled glass fiber composite. OHDMBSC. 2005;4(2):15-17,48-50.
15. Anusavice K. J. Phillips’ Science of Dental Materials. 11th edition. St. Louis: Saunders; 2003.
16. Venus H, Boening K, Peroz I. The effect of processing methods and acrylic resins on the accuracy of maxillary dentures and toothless denture bases: An in vitro study. Quintessence Int. 2011;42(8):669-677.